Characterization of DELUX: Ultraviolet light sterilization device for PFF2 / N95 masks against COVID-19
Main Article Content
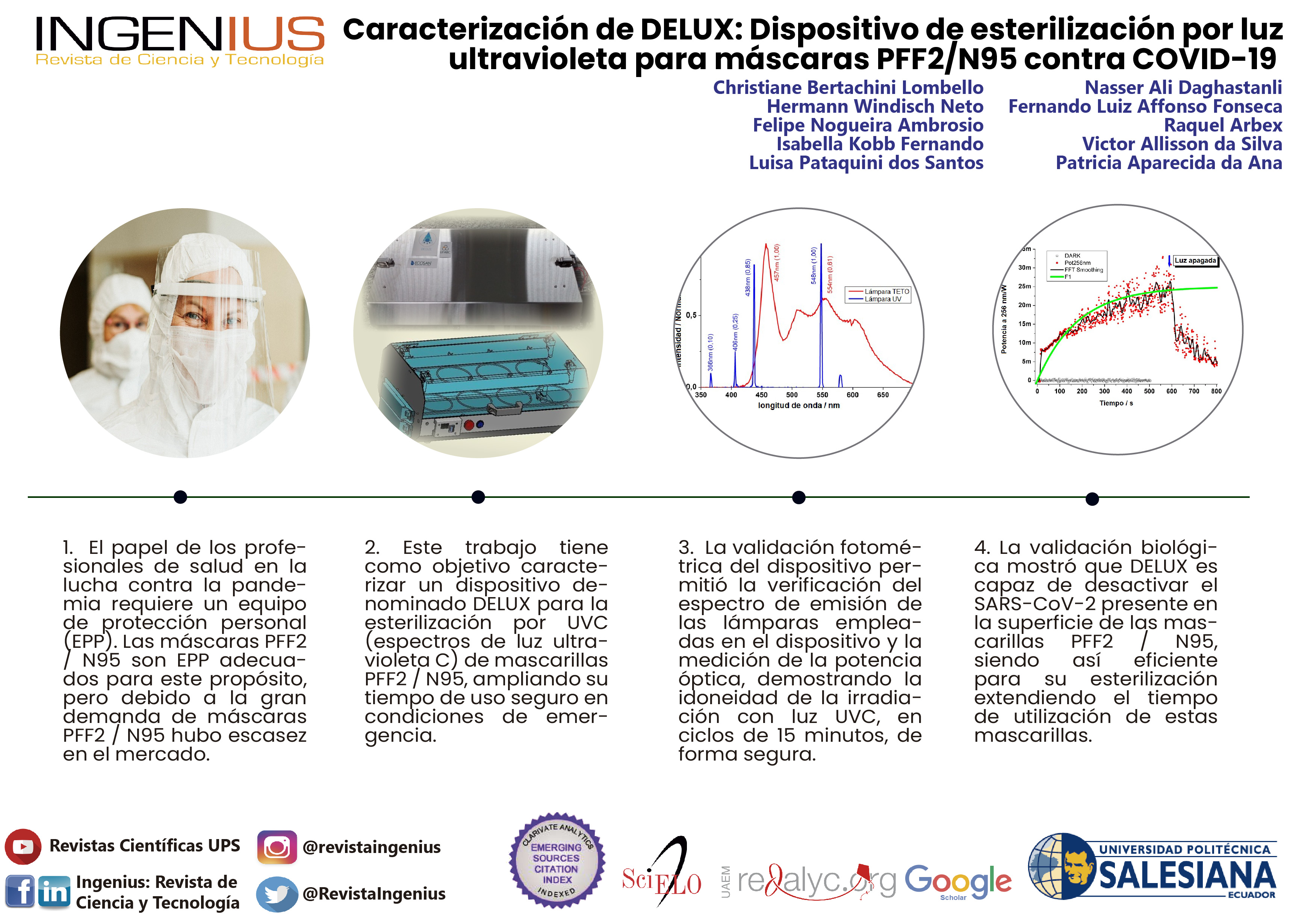
Abstract
Keywords
COVID-19, equipo, esterilización, protección, radiación, ultravioleta COVID-19, equipment, protective, radiation, sterilization, ultraviolet
References
[2] J. S. M. Peiris, “Coronaviruses,” Elsevier Public Health Emergency Collection, pp. 587–593. [Online]. Available: https://dx.doi.org/10.1016/B978-0-7020-4089-4.00072-X
[3] M. D. S. Cespedes and J. C. R. P. Souza, “Sars-cov-2: A clinical update - ii,” Revista da Associação Médica Brasileira, vol. 66, no. 4, pp. 547–557. [Online]. Available: https://doi.org/10.1590/1806-9282.66.4.547
[4] M. Pal, G. Berhanu, C. Desalegn, and V. Kandi, “Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2): An Update,” Pal M, Berhanu G, Desalegn C, Kandi V. Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2): An Update. Cureus. 2020;12(3):e7423. Published 2020 Mar 26. doi:10.7759/cureus.7423, vol. 12, no. 3, p. e7423, 2020. [Online]. Available: https://dx.doi.org/10.7759/cureus.7423
[5] D. X. Liu, J. Q. Liang, and T. S. Fung, “Human coronavirus-229e, -oc43, -nl63, and -hku1 (coronaviridae),” Elsevier Public Health Emergency Collection, pp. 428–440. [Online]. Available: https://dx.doi.org/10.1016/B978-0-12-809633-8.21501-X
[6] M. Wang, R. Cao, L. Zhang, X. Yang, J. Liu, M. Xu, Z. Shi, Z. Hu, W. Zhong, and G. Xiao, “Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro,” Cell research, vol. 30, no. 3, pp. 269–271, 2020. [Online]. Available: https://doi.org/10.34172/jrcm.2020.005
[7] S. H. Alfaraj, J. A. Al-Tawfiq, A. Y. Assiri, N. A. Alzahrani, A. A. Alanazi, and Z. A. Memish, “Clinical predictors of mortality of Middle East Respiratory Syndrome Coronavirus (MERS-CoV) infection: A cohort study,” Travel Medicine and Infectious Disease, vol. 29, pp. 48–50. [Online]. Available: https://doi.org/10.1016/j.tmaid.2019.03.004
[8] L. Piroth, J. Cottenet, A.-S. Mariet, P. Bonniaud, M. Blot, P. Tubert-Bitter, and C. Quantin, “Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide, population-based retrospective cohort study,” The Lancet Respiratory Medicine, vol. 9, no. 3, pp. 251–259, 2021. [Online]. Available: https://doi.org/10.1016/S2213-2600(20)30527-0
[9] FIOCRUZ. (2021) Por que a doença causada pelo novo coronavírus recebeu o nome de Covid-19? Fundação Oswaldo Cruz. [Online]. Available: https://bit.ly/3pwoiEE
[10] J. Zhang, W. Wu, X. Zhao, and W. Zhang, “Recommended psychological crisis intervention response to the 2019 novel coronavirus pneumonia outbreak in China: a model of West China Hospital,” Precision Clinical Medicine, vol. 3, no. 1, pp. 3–8, 02 2020. [Online]. Available: https://doi.org/10.1093/pcmedi/pbaa006
[11] M. Relvas, J. C. ao, R. Oliveira, J. C. Cardoso, and M. Gonçalo, “Manifestações cutaneas associadas a COVID-19: Uma revisão narrativa,” Revista Científica da Ordem dos Médicos, vol. 34, no. 2, pp. 128–136, 2021. [Online]. Available: https://doi.org/10.20344/amp.14574
[12] M. C. Grant, L. Geoghegan, M. Arbyn, Z. Mohammed, L. McGuinness, E. L. Clarke, and R. G. Wade, “The prevalence of symptoms in 24,410 adults infected by the novel coronavirus (SARS-CoV-2; COVID-19): A systematic review and meta-analysis of 148 studies from 9 countries,” PLoS One, vol. 15, no. 6, p. e0234765, 2020. [Online]. Available: https://doi.org/10.1371/journal.pone.0234765
[13] E. A. S. Medeiros, “A luta dos profissionais de sade no enfrentamento da COVID-19,” Acta Paul Enferm., p. 33, 2020. [Online]. Available: http://dx.doi.org/10.37689/acta-ape/2020EDT0003
[14] Secretaria de Vigiláncia em Saúde. (2021) Recomendações de proteção aos trabalhadores do serviço de saúde no contexto da pandemia da Covid-19. Ministério da Saúde, Brasil. [Online]. Available: https://bit.ly/3pvZ3lX
[15] C. D. Cappa, S. B. Sima Asadi, A. S. Wexler, N. M. Bouvier, and W. D. Ristenpart, “Expiratory aerosol particle escape from surgical masks due to imperfect sealing,” Scientific Reports, no. 11, p. 12110, 2021. [Online]. Available: https://doi.org/10.1038/s41598-021-91487-7
[16] ANVISA, Nota Técnica GVIMS/GGTES/ANVISA N? 04/2020. Orientações para serviços de saúde: medidas de prevenção e controle que devem ser adotadas durante a assistencia aos casos suspeitos ou confirmados de infecção pelo novo coronavirus (SARS-CoV-2). Agéncia Nacional de Vigiláncia Sanitária, 2021. [Online]. Available: https://bit.ly/3z12T9X
[17] ABNT, Equipamento de proteção respiratória – Filtros para partículas. Associação Brasileira de Normas Técnicas (ABNT), 2010. [Online]. Available: https://bit.ly/3qwTiUF
[18] ——, Equipamento de proteção respiratória – Peça semifacial filtrante para partículas. Associação Brasileira de Normas Técnicas (ABNT). [Online]. Available: https://bit.ly/3qqm09z
[19] IBSP. (2020) Máscaras n95 – recomenda˜ccões para uso prolongado e reutiliza˜ccão. Instituto Brasileiro Para Segurança Do Paciente. [Online]. Available: https://bit.ly/3Jpfd8D
[20] S. Duncan, P. Bodurtha, and S. Naqvi, “The protective performance of reusable cloth face masks, disposable procedure masks, kn95 masks and n95 respirators: Filtration and total inward leakage,” PLoS One, vol. 16, no. 10, p. e0258191, 2021. [Online]. Available: https://doi.org/10.1371/journal.pone.0258191
[21] C. C. Ontiveros, D. C. Shoults, S. MacIsaac, K. D. Rauch, C. L. Sweeney, A. K. Stoddart, and G. A. Gagnon, “Specificity of UV-C LED disinfection efficacy for three N95 respirators,” Scientific Reports, no. 11, p. 15350, 2021. [Online]. Available: https://doi.org/10.1038/s41598-021-94810-4
[22] D. B. Larremore, B. Wilder, E. Lester, S. Shehata, J. M. Burke, J. A. Hay, T. Milind, M. J. Mina, and R. Parker, “Test sensitivity is secondary to frequency and turnaround time for covid-19 surveillance,” medRxiv : the preprint server for health sciences, vol. 20, p. 20136309, Sep 2020. [Online]. Available: https://dx.doi.org/10.1101/2020.06.22.20136309
[23] CDC. (2020) Research Use Only 2019-Novel Coronavirus (2019-nCoV) Real-time RT-PCR Primers and Probes. Centers for Disease Control and Prevention. [Online]. Available: https://bit.ly/3EBvJyF
[24] T. I. C. on Non-Ionizing Radiation Protection, “Guidelines on limits of exposure to ultraviolet radiation of wavelengths between 180 nm and 400 nm (incoherent optical radiation),” Health Physics, vol. 87, no. 2, 2004. [Online]. Available: https://doi.org/10.1097/00004032-200408000-00006
[25] M. E. R. Darnell, K. Subbarao, S. M. Feinstone, and D. R. Taylor, “Inactivation of the coronavirus that induces severe acute respiratory syndrome, SARS-CoV,” J Virol Methods, vol. 121, no. 1, pp. 85–91, Oct. 2004. [Online]. Available: https://doi.org/10.1016/j.jviromet.2004.06.006
[26] K. Bedell, A. H. Buchaklian, and S. Perlman, “Efficacy of an automated multiple emitter whole-room ultraviolet-c disinfection system against coronaviruses mhv and mers-cov,” Infection Control & Hospital Epidemiology, vol. 37, no. 5, pp. 598–599, 2016. [Online]. Available: https://doi.org/10.1017/ice.2015.348
[27] F. A. Alexandre, J. A. F. Faria, and C. F. Cardoso, “Avaliação da eficiéncia da radiação ultravioleta na esterilização de embalagens plásticas,” Ciéncia e Agrotecnologia, vol. 32, no. 5, pp. 1524–1530, 2008. [Online]. Available: https://bit.ly/3mAbDie
[28] J. S. Smith, H. Hanseler, J. Welle, R. Rattray, M. Campbell, T. Brotherton, T. Moudgil, T. F. Pack, K. Wegmann, S. Jensen, J. Jin, C. B. Bifulco, S. A. Prahl, B. A. Fox, and N. L. Stucky, “Effect of various decontamination procedures on disposable n95 mask integrity and sars-cov-2 infectivity,” Journal of clinical and translational science, vol. 5, no. 1, pp. e10–e10, Jun 2020. [Online]. Available: https://dx.doi.org/10.1017/cts.2020.494
[29] M. Biasin, A. Bianco, G. Pareschi, A. Cavalleri, C. Cavatorta, C. Fenizia, P. Galli, L. Lessio, M. Lualdi, E. Tombetti, A. Ambrosi, E. M. A. Redaelli, I. Saulle, D. Trabattoni, A. Zanutta, and M. Clerici, “Uv-c irradiation is highly effective in inactivating sars-cov-2 replication,” Scientific Reports, vol. 11, no. 1, p. 6260, Mar 2021. [Online]. Available: https://doi.org/10.1038/s41598-021-85425-w
[30] CDC. Implementing Filtering Facepiece Respirator (FFR) Reuse, Including Reuse after Decontamination, When There Are Known Shortages of N95 Respirators. Centers for Disease Control and Prevention. [Online]. Available: https://bit.ly/3sD9ypQ
[31] K. O’Hearn, S. Gertsman, M. Sampson, R. Webster, A. Tsampalieros, R. Ng, J. Gibson, A. T. Lobos, N. Acharya, A. Agarwal, S. Boggs, G. Chamberlain, E. Staykov, L. Sikora, and J. D. McNally, “Decontaminating n95 and sn95 masks with ultraviolet germicidal irradiation does not impair mask efficacy and safety,” vol. 106, no. 1, pp. 163–175. [Online]. Available: https://doi.org/10.1016/j.jhin.2020.07.014
[32] L. Liao, W. Xiao, M. Zhao, X. Yu, H. Wang, Q. Wang, S. Chu, and Y. Cui, “Can n95 respirators be reused after disinfection? how many times?” ACS nano, vol. 14, no. 5, pp. 6348–6356, May 2020. [Online]. Available: https://dx.doi.org/10.1021/acsnano.0c03597
[33] W. G. Lindsley, S. B. Martin, Jr, R. E. Thewlis, K. Sarkisian, J. O. Nwoko, K. R. Mead, and J. D. Noti, “Effects of ultraviolet germicidal irradiation (UVGI) on N95 respirator filtration performance and structural integrity,” J Occup Environ Hyg, vol. 12, no. 8, pp. 509–517, 2015. [Online]. Available: https://doi.org/10.1080/15459624.2015.1018518
[34] ABMT. 87 % das denúncias de falta de EPIs são sobre máscaras N95, diz AMB. Associação Brasileira de Medicina do Trabalho. [Online]. Available: https://bit.ly/3JlBFj5
[35] J. Rose. (2020) Why can’t america make enough n95 masks? 6 months into pandemic, shortages persist. NPR. SPECIAL SERIES The Coronavirus Crisis. [Online]. Available: https://n.pr/3mBjImT
[36] K. Seresirikachorn, V. Phoophiboon, T. Chobarporn, K. Tiankanon, S. Aeumjaturapat, S. Chusakul, and K. Snidvongs, “Decontamination and reuse of surgical masks and N95 filtering facepiece respirators during the COVID-19 pandemic: A systematic review,” Infect Control Hosp Epidemiol, vol. 42, no. 1, pp. 25–30, Jul. 2020. [Online]. Available: https://doi.org/10.1017/ice.2020.379
[37] E. C. Toomey, Y. Conway, C. Burton, S. Smith, M. Smalle, X.-H. S. Chan, A. Adisesh, S. Tanveer, L. Ross, I. Thomson, D. Devane, and T. Greenhalgh, “Extended use or reuse of single-use surgical masks and filtering face-piece respirators during the coronavirus disease 2019 (COVID-19) pandemic: A rapid systematic review,” Infect Control Hosp Epidemiol, vol. 42, no. 1, pp. 75–83, Oct. 2020. [Online]. Available: https://doi.org/10.1017/ice.2020.1243
[38] NEVMS, Descontaminação de máscaras de proteção respiratória modelo N95 (respirador particulado). Núcleo de Evidéncia de Mato Grosso do Sul, 2020. [Online]. Available: https://bit.ly/345EmW1
[39] B. K. Heimbuch, W. H. Wallace, K. Kinney, A. E. Lumley, C.-Y. Wu, M.-H. Woo, and J. Wander, “A pandemic influenza preparedness study: use of energetic methods to decontaminate filtering facepiece respirators contaminated with H1N1 aerosols and droplets,” Am J Infect Control, vol. 39, no. 1, pp. e1–9, Dec. 2010. [Online]. Available: https://doi.org/10.1016/j.ajic.2010.07.004
[40] S. Sangkham, “Face mask and medical waste disposal during the novel covid-19 pandemic in asia,” vol. 2, p. 100052. [Online]. Available: https://doi.org/10.1016/j.cscee.2020.100052